Bcl-2 proteins are a family of homo- or hetero-dimeric signalling proteins that are involved in apoptosis regulation. The Bcl-2 (B-cell lymphoma 2) protein itself is an important therapeutic drug target for cancer, more specifically: CLL (chronic lymphocytic leukemia). Mutations in the BCL2 gene are known to be the cause of various cancers, cancer resistance, and autoimmune diseases.
Bcl-2 acts as an anti-apoptotic (pro-survival) signal by binding (heterodimerizing) and inhibiting pro-apoptotic proteins including Bax and Bak (among numerous other binding partners). Bax and Bak themselves are two pro-apoptotic Bcl2-family (Bcl-2-like) proteins and are able to dimerize with Bcl-2 via Bcl-2-homology (BH) domains.
Shown below is a structure of Bcl-2 bound to a segment of the BH3 domain of Bax (PDB 2XA0).
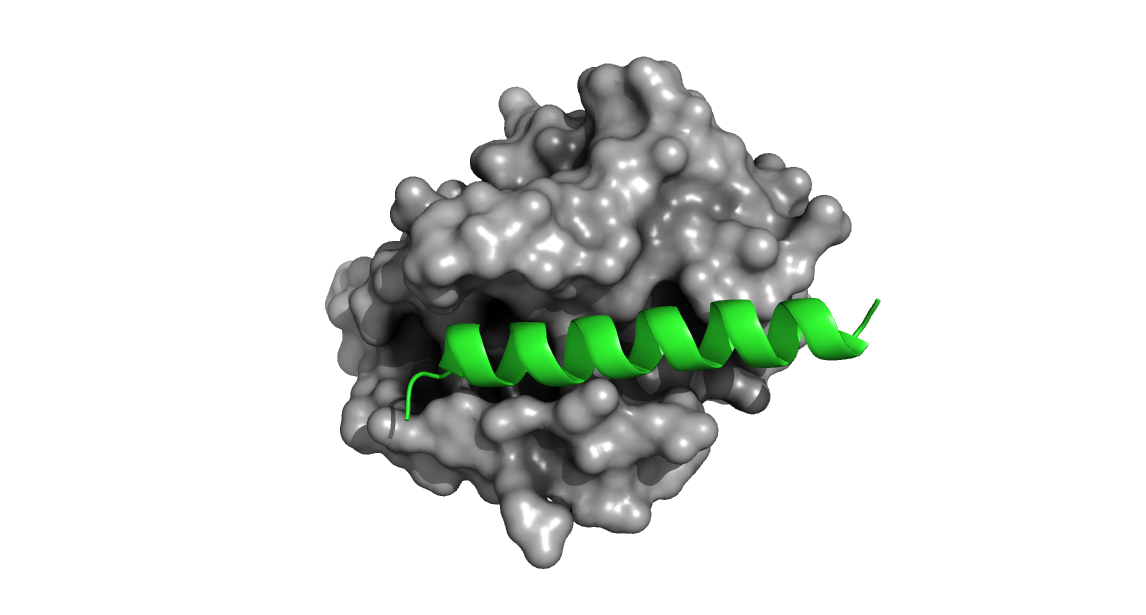
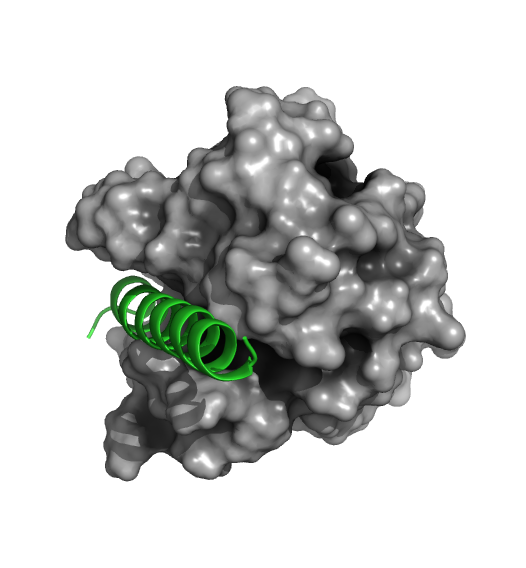
Figure 1. Bcl-2 (grey) bound with a segment of the Bax BH3 domain (green). Note how the BH3 peptide engages the groove of Bcl-2.
Venetoclax is a selective inhibitor of Bcl-2 and this drug is notable in the field of fragment-based drug discovery in that it was the second approved drug to be developed using this approach.
The figure below shows that venetoclax engages the same pocket that is normally the binding site for BH3 domains on Bcl-2 binding partners.
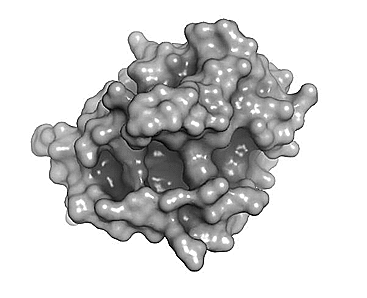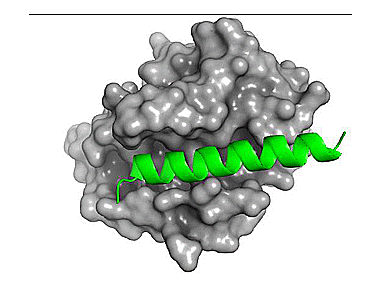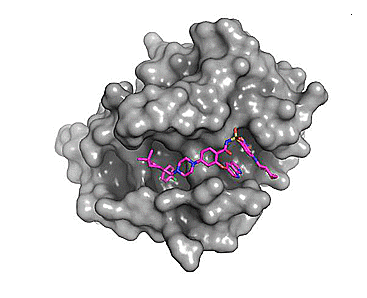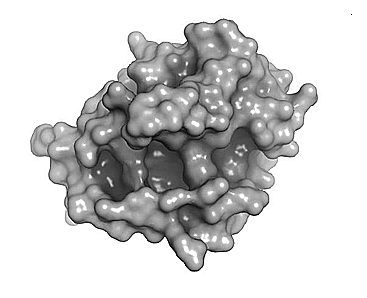
Figure 2. Comparison of the binding site for the small molecule inhibitor, venetoclax and a native BH3 peptide (PDB: 6O0P and 2XA0, respectively).