Lysozyme - antimicrobial enzyme part of the innate immune system
Lyzozymes are a family of enzymes that cleave (via hydrolysis)
peptidoglycans (polymers of sugar and amino acids). Because
peptidoglycan is a large component of bacterial cell walls, lysozyme
proteins can thereby act as an antimicrobial by destabilizing bacterial
cell integrity.
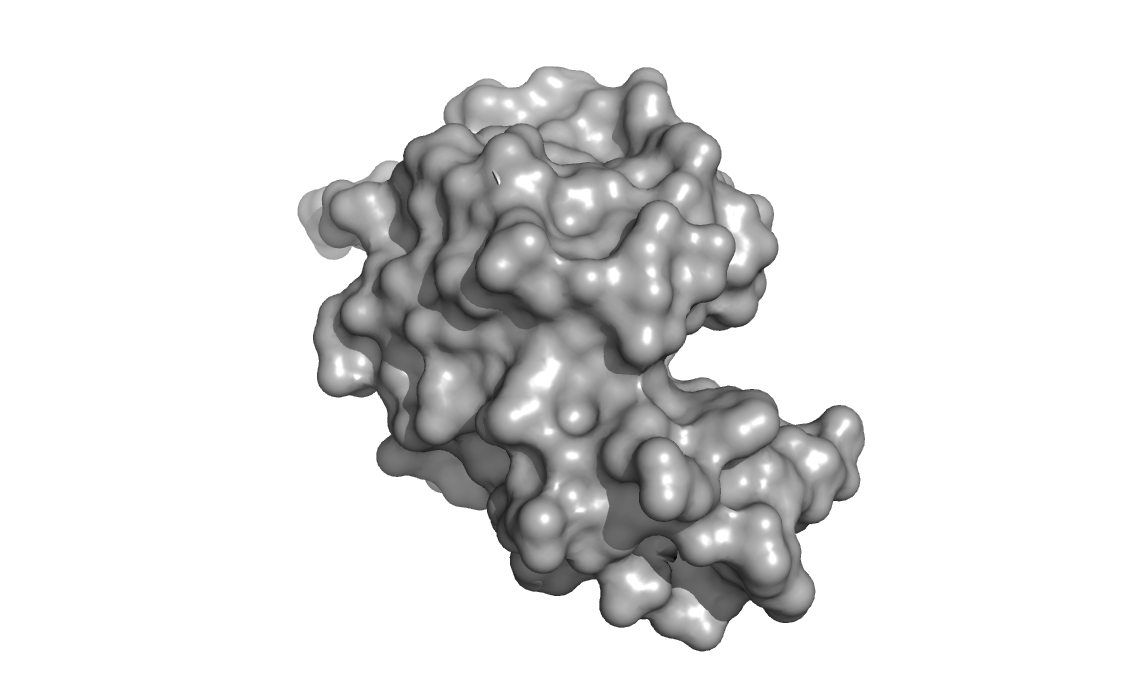
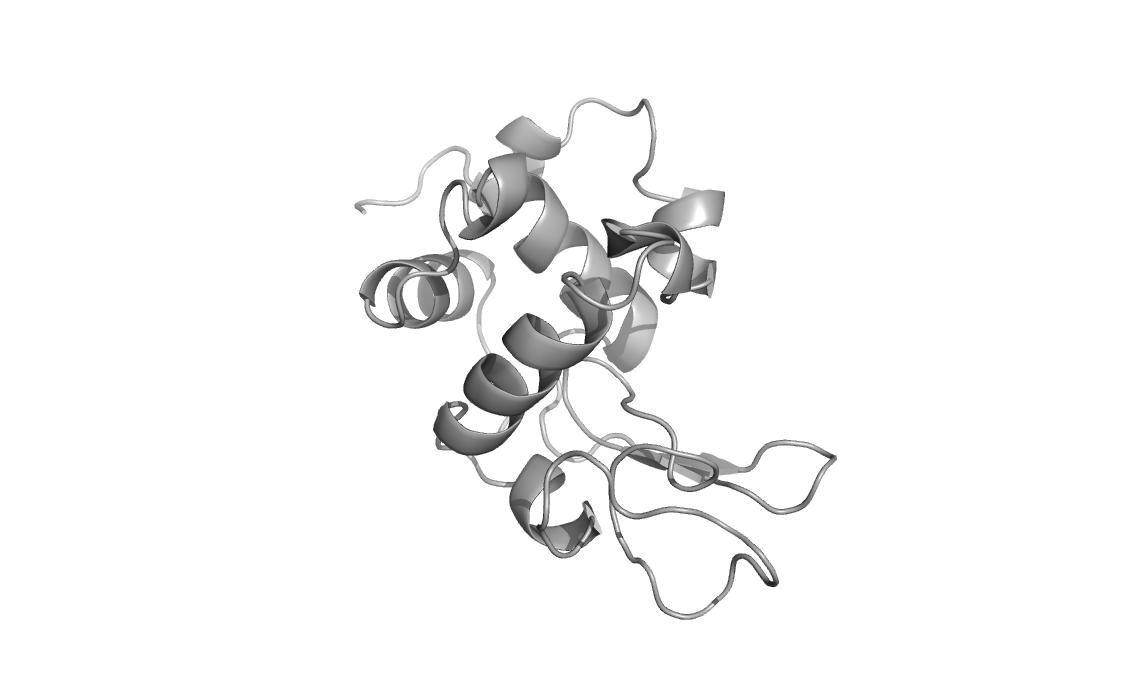
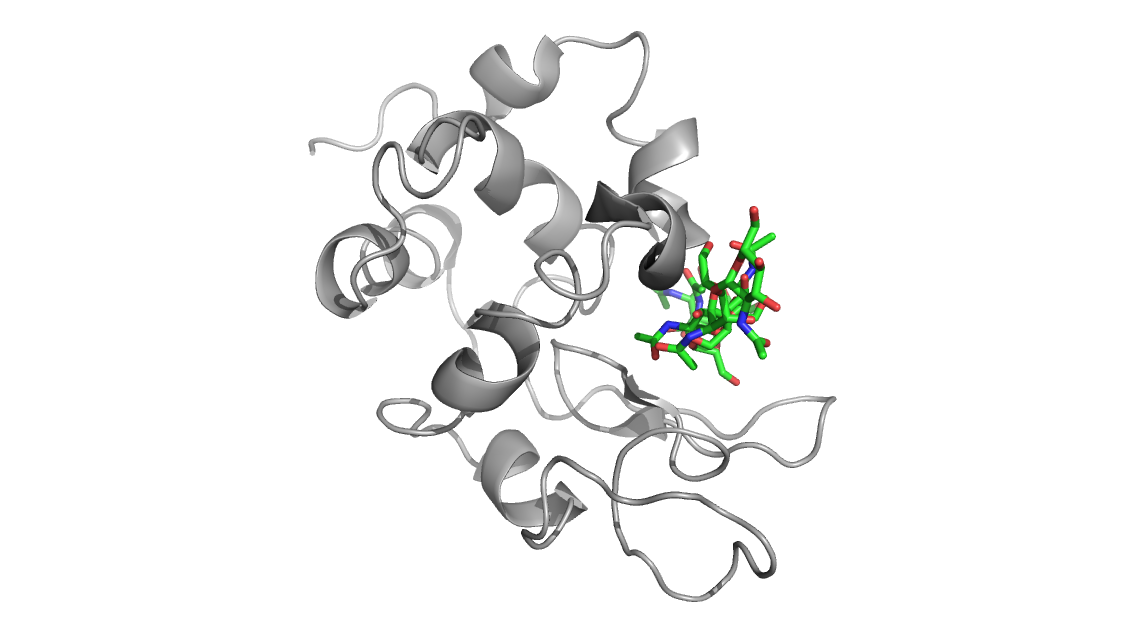
Above are three representations of egg white lysozyme (PDB: 193L and
PDB: 1SFG). Notice the large cleft on the right side of the protein.
This is where peptidoglycan (represented in green) inserts and is
positioned for catalytic hydrolysis (third image on right).

Lysozyme is found in many animal secretions, including: tears, saliva,
milk, mucus, and egg whites. It has been routinely used as a model
protein for many studies in biochemistry and structural biology in
large part due to the ease of isolation, thermal and pH stability, and
quantities available from egg whites. Its structure was the first of
any enzyme to be solved by x-ray crystallography techniques.
Below is a natural abundance 15N-HSQC of egg white lysozyme recorded on a 600 MHz NMR.
Here is a two-dimensional carbon HSQC spectrum of reductively
methylated egg-white lysozyme. The reductive methylation approach has
been used by crystallographers to occasionally improve the
crystallization properties of otherwise difficult to crystallize
proteins. The applications of this methylation technique also extend to
spectroscopic fingerprinting of conformational states. Each methyl
group gives rise to peaks in the spectrum that correspond to the
microenvironment of N-terminal amino groups and lysine amino groups in
proteins. Given that salt-bridges are often found in proteins to
stabilize key interactions, addition of a methyl probe allows us to
monitor salt-bridge stability, state (on-off), pH-dependence, and
global conformational changes in labeled proteins.
Cryo Electron Microscopy (Cryo-EM) is a technique that has been a staple in the field of structural biology for some time. Significant improvements have been made in single
particule cryoEM and as a result it has been attracting a lot of attention. Cryo-EM has been used for structural
determination of large proteins and protein complexes, but for a long time, the technique
failed to achieve atomic resolution comparable to X-ray
crystallography / diffraction techniques. However, it is now routinely being used to solve structures of
large proteins and their complexes at high resolution.
A final limitation has been overcome in that whereas previously, CryoEM could only be used to study large proteins, it can now be used to generate high
resolution structures of small proteins. Micro Electron Diffraction (MicroED) can be used to
resolve small protein structures (such as lysozyme, 14 kDa) with resolution
comparable to x-ray diffraction.
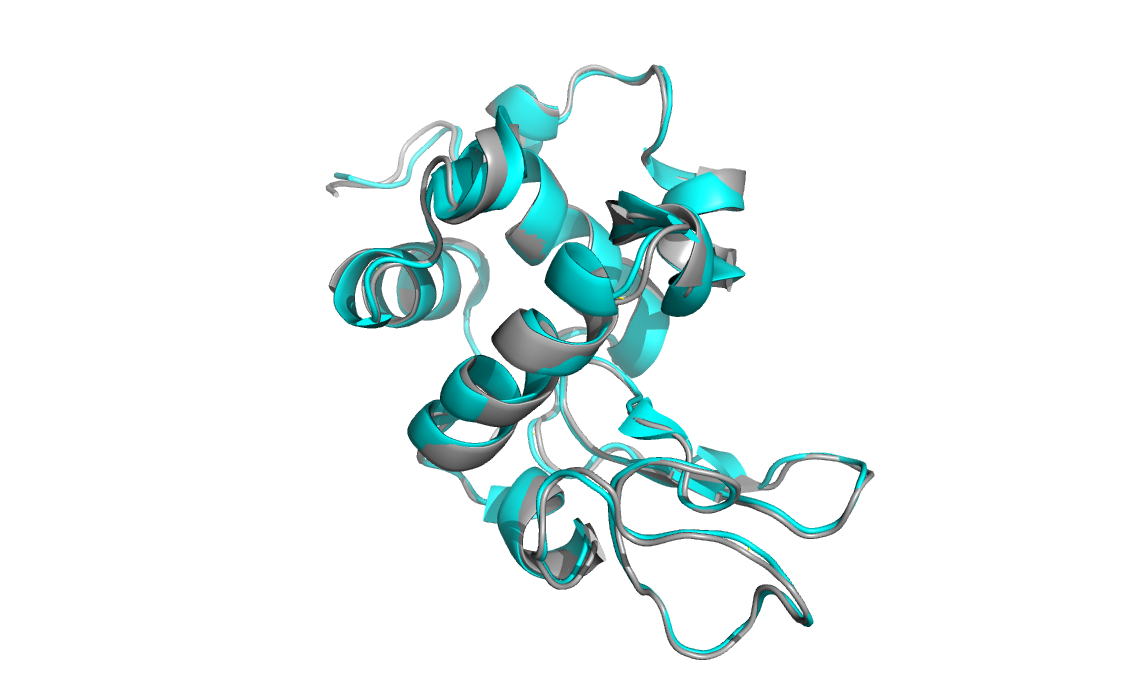
Below is a microED structure of lysozyme solved to 2.9 angstroms
(cyan, PDB: 3J4G) aligned to an x-ray crystallographic structure of the
same protein (grey, PDB:193L). The backbone structures are nearly
identical.
Main Page




